 Autophagy is a way that cells can survive low oxygen / nutrient conditions and other kinds of stress. The autophagy pathway can be activated also when tumor cells are stressed by chemotherapy or radiation. Autophagy is a way that cells can survive low oxygen / nutrient conditions and other kinds of stress. The autophagy pathway can be activated also when tumor cells are stressed by chemotherapy or radiation.
Whether activation of autophagy allows tumor cells to survive therapy or is a desirable mechanism for initiating a non-apoptotic form of programmed cell death remains controversial PubMed. It may depend on the type of cancer or perhaps on the mutations that are driving its malignant behavior.
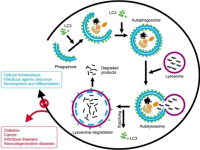
Autophagy (technical definition): is a process of self-cannibalization. Cells capture their own cytoplasm and organelles and consume them in lysosomes. The resulting breakdown products are inputs to cellular metabolism, through which they are used to generate energy and to build new proteins and membranes. Autophagy preserves the health of cells and tissues by replacing outdated and damaged cellular components with fresh ones. In starvation, it provides an internal source of nutrients for energy generation and, thus, survival. A powerful promoter of metabolic homeostasis at both the cellular and whole-animal level, autophagy prevents degenerative diseases. It does have a downside, however—cancer cells exploit it to survive in nutrient-poor tumors. http://www.ncbi.nlm.nih.gov/pmc/articles/PMC3010857/
Based on recent findings it has been hypothesized that agents that block autophagy may sensitize cancer cells to regular treatments. One such agent is chloroquine (a drug used to treat malaria). The testing of the treatment-sensitizing properties of chloroquine are being evaluated in humans cancers. See Clinicaltrials.gov
Here we ask if this approach is promising also for the treatment of some types or variants of lymphoma. What follows is background on the autophagy pathway and the rationale for testing agents that prevent cell survival by autophagy in combination with regular treatments.
Autophagy Reports
.gif) |
Autophagy inhibition enhances therapy-induced apoptosis in a Myc-induced model of lymphoma http://1.usa.gov/1oHqO6w
Accumulating evidence suggests that autophagy can also represent an adaptive strategy by which cells clear damaged organelles and survive bioenergetic stress. Autophagy, by targeting cytoplasmic proteins and organelles for lysosomal degradation, plays a role in recycling organelles and proteins that may be damaged by increased reactive oxygen species generated by the cellular stress associated with activated oncogenes and cancer therapies (7, 8). Autophagy also promotes the survival of cells resistant to apoptosis when they are deprived of extracellular nutrients or growth factors. Treatment of such cells dependent on autophagy for survival with the drug chloroquine (CQ) results in brisk cell death (9).
These studies provide evidence that autophagy serves as a survival pathway in tumor cells treated with apoptosis activators and a rationale for the use of autophagy inhibitors such as chloroquine in combination with therapies designed to induce apoptosis in human cancers."
|
.gif) |
Overexpression of LC3A autophagy protein in lymphoma [Hematol Oncol Stem Cell Ther. 2013] - PubMed - NCBI http://1.usa.gov/1oK8Pg0
|
.gif) |
EJP, 2009: Chloroquine and its analogs: a new promise o... [Eur J Pharmacol. 2009] - PubMed - NCBI http://1.usa.gov/1pFwWvi
|
.gif) |
Autophagy inhibition in combination cancer treatment [Curr Opin Investig Drugs. 2009] - PubMed - NCBI http://1.usa.gov/1oURVFA
The effective elimination of cancer cells is compromised by mechanisms of resistance.
Such mechanisms have been variably ascribed to drug export transporters, more effective DNA repair mechanisms compared with healthy cells, singularly resistant stem cells, resistance to apoptosis, self-sufficiency for growth factor signaling and an angiogenic switch, as well as immunological pathways associated with T-regulatory cells, myeloid-derived suppressor cells or plasmacytoid dendritic cells.
In this review, the critically important process of autophagy, which is a mechanism of cell survival in the presence of genomic injury, endoplasmic reticulum stress, oxidant stress, nutrient insufficiency and viral/bacterial infection, is explored in the setting of cancer treatment. Autophagy has recently been demonstrated as important for conferring resistance to chemotherapy, radiation therapy and immunotherapy.
Compounds are now available that can reverse autophagy, including the antimalarial compounds chloroquine and hydroxychloroquine, as well as the antidepressant agent clomipramine. Other strategies for the reversal of autophagy are based on the recent observation that the cytosolic location of the chromatin-binding protein HMGB1 (high-mobility group box-1) is associated with sustained autophagy. Targeting HMGB1 using platinum-containing compounds, ethyl pyruvate or glycyrrhizin has also been used to limit autophagy. Screening for new agents is ongoing, which, coupled with conventional chemotherapeutic compounds, may usher in a new generation of autophagy-inhibiting agents.
|
.gif) |
ScienceDirect: Growth Factor Regulation of Autophagy and Cell Survival in the Absence of Apoptosis http://bit.ly/1mcWbR0
|
|