Topics
Overview of epigenetic therapy:
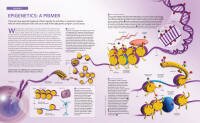 It's well established that cancer arise from genetic mutations. However, so-called epigenetic mechanisms also drive the malignant behavior - by turning OFF or ON normal genes that can control the growth and persistence of the malignant cells. It's well established that cancer arise from genetic mutations. However, so-called epigenetic mechanisms also drive the malignant behavior - by turning OFF or ON normal genes that can control the growth and persistence of the malignant cells.
For example, tumor suppressor genes that would normally start the cell death process when a cell becomes defective, can be turned off in the malignant cells. Put a little differently, whether a cell becomes cancerous depends not only mutations to key genes (the genome), but also whether or not other key genes (un-mutated) are expressed appropriately (the epigenome).
The mutations in genes can affect the expression of other genes. Epigenetic "levers" (methylation and acetylation) can be targeted by a relatively new class of treatment agents to control the expression of genes – without affecting the mutated DNA sequence itself.
Epigenetic agents have become the focus of a rapidly emerging and important new area of cancer research. These are compounds that modify gene expression by methylation or acetylation of histones and other elements such as MicroRNA in order reprogram how the tumor cells behave: grow, die, or differentiate.
See also for our hand-picked background videos on the Epigenetics:
Epigenetics 101 - Paul Andersen explains the concepts 
The Epigenome at a Glance 
Histone deacetylase inhibitors
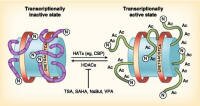 Histone deacetylase (HDACs) inhibitors appear to be active against cancer cells by "loosening the grip" of histones on genes within the malignant cell - restarting tumor suppressor genes (built in protections against cancer within the cells) helping to cause the tumor cells to die or to stop growing. Histone deacetylase (HDACs) inhibitors appear to be active against cancer cells by "loosening the grip" of histones on genes within the malignant cell - restarting tumor suppressor genes (built in protections against cancer within the cells) helping to cause the tumor cells to die or to stop growing.
In short, HDACs are thought to work by restoring the capability of malignant cells turn on genes that help the malignant cells to undergo programmed cell death. I think of histones as string around a trigger finger. When the string is wound too tightly, the finger cannot pull the trigger that initiates cell death. Agents that loosen the string can allow genes that provide these defenses to do their job.
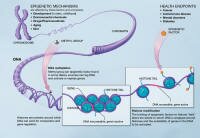
DNA hypo/hyper-methylation agents
These are agents that can reactivate tumor suppressor genes by altering methylation.
Hypermethylation-associated silencing of tumor suppressor genes "has been shown to occur in lymphoid/hematopoietic malignancies, disrupting many cellular pathways. The cytidine analogs 5-aza-2′-deoxycytidine and 5-azacytidine are capable of reactivating tumor suppressor genes that were silenced by hypermethylation.4
In the News
.gif) |
Abexinostat (PCI-24781) (pan HDAC inhibitor) Find trials
|
.gif) |
|
.gif) |
* Weil Cornell 2014:
FDA Announces Approval for Beleodaq (belinostat) in Treatment of Peripheral T-cell Lymphoma FDA Announces Approval for Beleodaq in Treatment of Peripheral T-cell Lymphoma http://bit.ly/1t6j54x
“The safety and effectiveness of Beleodaq was evaluated in a clinical study involving 129 participants with relapsed or refractory PTCL. All participants were treated with Beleodaq until their disease progressed or side effects became unacceptable. Results showed 25.8 percent of participants had their cancer disappear (complete response) or shrink (partial response) after treatment.”
belinostat A novel hydroxamic acid-type histone deacetylase (HDAC) inhibitor with antineoplastic activity. Belinostat targets HDAC enzymes, thereby inhibiting tumor cell proliferation, inducing apoptosis, promoting cellular differentiation, and inhibiting angiogenesis.
This agent may sensitize drug-resistant tumor cells to other antineoplastic agents, possibly through a mechanism involving the down-regulation of thymidylate synthase. C
|
.gif) |
ASCO 2014:
Responses to Romidepsin by line of therapy in relapsed/refractory peripheral T-cell lymphoma http://bit.ly/1gBq3sO
|
.gif) |
* 2014, Sonali Smith, Blood:
Honing in on the (epi)genetic basis of AITL http://bit.ly/1mZ55XL
In this issue of Blood, Odejide et al report on recurring mutations in a large series of patients with angioimmunoblastic T-cell lymphoma (AITL) clustering around three epigenetic modifiers: TET2, DNMT3, and IDH2.1
|
.gif) |
BJM 2014:
Report on Vorinostat in relapsed indolent B-cell non-Hodgkin lymphoma and MCL - http://bit.ly/1juoJZY
Histone deacetylase (HDAC) inhibitors, such as vorinostat are thought to exhibit anti-tumour activity by up-regulating tumour-suppressive genes, promoting cell differentiation and inducing cell apoptosis. Vorinostat is an oral HDAC inhibitor approved for the treatment of refractory cutaneous T-cell lymphoma (CTCL; Olsen et al, 2007).
"In conclusion, this phase II study demonstrated the promising efficacy and safety of vorinostat in patients with relapsed or refractory FL. As this was a single-arm study with limited data to interpret, a comparative study to verify the efficacy of vorinostat is warranted. Given the less toxic profile of vorinostat, this drug could possibly be combined with rituximab or other molecular targeted drugs or could be used for maintenance after rituximab-containing chemotherapy in FL patients. Further clinical studies are warranted to elucidate the drug efficacy of vorinostat.
PAL query: List lymphoma trials for this agent: http://bitly.com/aFsxvB
|
.gif) |
Phase I/II Trial of Azacytidine + R-CHOP in Diffuse Large B-Cell Lymphoma http://bit.ly/ksAwY9
|
.gif) |
Cancer Discov. 2013
Mechanism-based epigenetic chemosensitization in lymphoma ... - PubMed http://1.usa.gov/1aa2npR
|
.gif) |
Bringing Epigenetic Therapy to the Forefront of Cancer Management http://bit.ly/1a8XDBp
|
Epigenetic agents in clinical phase testing
® indicates agents approved for lymphoma
Agent
|
|
Target
|
|
Find Trials
|
Last count of clicks |
Reports |
|
Vidaza ®
(5-Azacytidine)
|
|
DNA hypo-methylation |
|
Find trials
> |
20 |
Reports
Phase I/II Trial of Azacytidine + R-CHOP |
Abexinostat (PCI-24781)
|
|
HDAC |
|
Find trials
> |
25 |
Reports |
Beleodaq® Belinostat
(PXD101)
|
T or B |
HDAC |
|
Find trials
> |
32 |
Reports |
Mocetinostat
|
|
HDAC |
|
Find trials |
New |
Reports
> |
Panobinostat (LBH589)
|
T or B |
HDAC |
|
Find trials
> |
54 |
Reports |
Romidepsin® (Depsipeptide)
|
T or B |
HDAC |
Approved for T-cell lymphoma |
Find trials
> |
21 |
Reports |
Valproic acid
|
T or B |
HDAC |
AML / MDS |
Find trials
> |
78 |
Reports |
Zolinza ® (Vorinostat)
|
T or B |
HDAC |
Approved for T-cell lymphoma |
Find trials
> |
87 |
Reports |
| |
under development |
|
New HDAC agents
Testing Mocetinostat (HDAC inhibitor) with Brentuximab Vedotin in Patients With Relapsed or Refractory Hodgkin Lymphoma http://1.usa.gov/1Q6Wf4W
mocetinostat
A rationally designed, orally available, Class 1-selective, small molecule, 2-aminobenzamide HDAC inhibitor with potential antineoplastic activity. Mocetinostat binds to and inhibits Class 1 isoforms of HDAC, specifically HDAC 1, 2 and 3, which may result in epigenetic changes in tumor cells and so tumor cell death; although the exact mechanism has yet to be defined, tumor cell death may occur through the induction of apoptosis, differentiation, cell cycle arrest, inhibition of DNA repair, upregulation of tumor suppressors, down regulation of growth factors, oxidative stress, and autophagy, among others. Overexpression of Class I HDACs 1, 2 and 3 has been found in many tumors and has been correlated with a poor prognosis.
Trials of Interest:
.gif) |
DLBCL including transformed
relapsed or refractory AND not eligible for transplant
Testing: 5-azacitidine (epigenetic agent) WITH Vorinostat (HDAC inhibitor)
Location: Weill Cornell Medical College New York, New York, United States. Open study above for details.
|
.gif) |
DLBCL previously untreated
Testing: 5-azacitidine (epigenetic) WITH R-CHOP (standard therapy)
Location: Weill Cornell Medical College New York, New York, United States. Open study above for details.
Recruitment status unknown
Study drug reports Positive interim reports published recently for initial participants.
|
Resources and Reports
.gif) |
Scientists just made the first map of the human epigenome. The Washington Post http://wapo.st/17joH2e
See also background on epigenome http://www.genome.gov/27532724
Each person's body contains trillions of cells, all of which have essentially the same genome. Yet some cells are optimized for use in muscles, others for bones, the brain, the stomach and the rest of your body. What makes these cells different?
The protein-coding parts of your genome, called genes, do not make proteins all of the time in all of your cells. Instead, different sets of genes are turned on or off in various kinds of cells at different points in time. Differences in the types and amounts of proteins produced determine how cells look, grow and act. The epigenome influences which genes are active - and which proteins are produced - in a particular cell.
|
.gif) |
Rita Shaknovich and Ari Melnick 2010: Epigenetics and B-cell Lymphoma http://1.usa.gov/1aqVz7R
Mutations affecting epigenetic and transcriptional modifiers now appear to be an almost universal feature of B-cell lymphomas. Large-scale disruptions of DNA methylation and histone modification patterning are emerging as a hallmark of these diseases. The challenge for the next years will be to understand the nature of the lymphoma epigenome and the biochemical and biological effects of mutant epigenetic factors. It will also be necessary to ascertain the utility of DNA methylation classifiers as biomarkers for diagnostic accuracy and therapeutic stratification. Because epigenetic marks are potentially reversible, the development of genuine epigenetic-targeted therapy drugs holds great promise. Already agents are emerging that target transcription factors, chromatin modifying enzymes and epigenetic reader proteins (e.g. [49][50]). In the long-term such agents might significantly advance the treatment of B-cell lymphomas with less toxicity to the normal immune system and other tissues.
|
.gif) |
Epigenetic expression as biomarker:
Identification of Highly Methylated Genes across Various Types of B-Cell Non-Hodgkin Lymphoma http://1.usa.gov/1bH9Lph
|
.gif) |
Characteristics of HDACs in trials - a table:
http://www.ncbi.nlm.nih.gov/pmc/articles/PMC3100649/table/tab1/
Cell death is broadly divided into three forms - and many targeted drugs work by activating apoptosis and autophagy:
apoptosis - A type of cell death in which a series of
molecular steps in a cell leads to its death. This is
the body's normal way of getting rid of unneeded or
abnormal cells. The process of programmed cell death
may be blocked in cancer cells.
autophagy - A normal process in which a cell destroys
proteins and other substances in its cytoplasm (the
fluid inside the cell membrane but outside the
nucleus), which may lead to cell death.
necrosis - The process (not induced by HDACs) occurring
in cells that are dying from injuries that cannot be
fixed by the cell. It is characterized by swelling and
cell membrane rupture. Distinguish it from apoptosis
which is a normal, regulated cellular process.
|
.gif) |
|
.gif) |
|
.gif) |
Epigenetics 101 - Paul Andersen explains the concepts. http://bit.ly/1iDWHJK
|
.gif) |
The Epigenome at a Glance - YouTube http://bit.ly/1lithjY
|
|