|
TOPICS
Background Resources | Topic Searches | Clinical Trials | Treatment Updates | PET for T-cell lymphoma?
In the News
Introduction
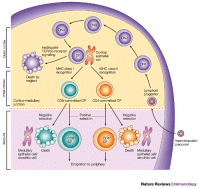 Lymphoma is a blood cell cancer - a condition where abnormal lymphocytes (a type of white blood cell) expand in number forming tumors often in lymph nodes but also in other regions, such as the bone marrow. See lymphatic system and Lymphoma Simplified. Lymphoma is a blood cell cancer - a condition where abnormal lymphocytes (a type of white blood cell) expand in number forming tumors often in lymph nodes but also in other regions, such as the bone marrow. See lymphatic system and Lymphoma Simplified.
Peripheral T-cell lymphomas describes a diverse group of blood cancers that originate from T-cells, which may be at various stages of development.
These lymphomas usually presents at diagnosis in stage III or IV, and often has an aggressive clinical course requiring prompt treatment.
Histologies include (non-cutaneous):
.gif) |
PTCL = peripheral t-cell lymphoma, NOS = Not otherwise specified
|
.gif) |
AITL = Angioimmunoblastic T-cell lymphoma
|
.gif) |
ALCL = Anaplastic large cell lymphoma
|
.gif) |
Enteropathy associated T-cell lymphomas
|
.gif) |
NK lymphoma (NK = Natural Killer Cell)
Source: NCCN.org PDF (login req.)
|
Incidence: More frequent in adults than children. Low incidence - approximately 10-15% of non-Hodgkin's lymphoma. Account for about 12% of lymphoid tumours worldwide. Uncommon in North America. Peripheral T-Cell Lymphoma Unspecified is the most common group. Also see specific variants (subtypes), below.
Grade: aggressive clinical course.
Staging: Staging refers to the how widespread the disease is. Imaging tests (CT MRI, PET, Gallium). See Staging for more detail.
Diagnosis requires analysis of tissue sample using a variety of tests to identify the cell type. T-cell markers will "express CD4 or CD8, but not both." 1
Prognosis: Each lymphoma is unique and the prognosis can depend on many clinical factors, including the unique molecular and biological characteristics of the tumor, the patient's age, general health and immune status, areas of involvement, how widespread the disease is at diagnosis (stage), and if so-called b-symptoms are present.
Also see Prognostic Markers for articles on biomarkers that may predict risk of disease
Treatment: Combination chemotherapy, such as CHOP, followed by stem cell transplantation. For investigational approaches, see below.
"Although CHOP remains the standard front-line therapy for PTCL, given these poor outcomes, most PTCL patients (with the exception of ALK-positive ALCL) should be considered for clinical trials of initial therapy." 2
Return to top
PET for aggressive T-cell lymphoma?
* ASH Paper:
Negative Interim FDG-PET Scan Is Predictive of Superior Outcome in T Cell Lymphoma http://bit.ly/1iDzGpb
* Prognostic Value of Interim and Posttherapy 18F-FDG PET/CT in Patients with Mature T-Cell and Natural Killer Cell Lymphomas http://bit.ly/QsitVu
Our study indicates that interim and posttherapy PET/CT results are 2 independent predicators of PFS and OS in T/NK-cell lymphomas. In addition, our data also imply that patients with positive results at interim or post therapy PET/CT should be considered candidates for an intensive therapeutic strategy to improve their clinical outcome.
References and News
Re: Blood 2014: Alison J. Moskowitz, Matthew A. Lunning and Steven M. Horwitz
How I treat the peripheral T-cell lymphomas http://bit.ly/1k8AW1Q
Technical -- but for anyone diagnosed with PTCL ... a very important paper to look over and share with our oncologists - underlining the potential importance of consulting specialists in this area.
One of the more challenging aspects in managing PTCL is confidently arriving at the diagnosis. T-cell lymphomas are rare, and we try to approach new diagnoses with at least a little skepticism. For many B-cell lymphomas, reproducible immunophenotypic patterns and cytogenetic features allow for an algorithmic approach to diagnosis. On the contrary, T-cell lymphomas are often characterized by antigen aberrancy that may vary within a subtype or even over the course of disease.7,8
The most powerful predictor of outcome in our series was interim PET; 53% of patients normalized their PET after 4 cycles of chemotherapy and in those who achieved interim PET-negative status, 59% were progression free at 5 years, including 53% of those with IPI of ≥3.
Our preference for fit patients is for alloSCT in the relapsed/refractory setting. Our institutional data and others have shown that the use of autologous stem cell transplantation for relapsed PTCL, with a possible exception of ALCL, has rarely resulted in long-term disease control.40,41 This is somewhat controversial and a recent analysis from the Center for International Blood and Marrow Transplant Research registry points to better results with ASCT at relapse, although the series is overrepresented by ALCL including many with ALK-positive ALCL.42
* Br J Haematol 2013:
A phase I/II trial of bortezomib combined concurrently with gemcitabine for relapsed or refractory DLBCL and peripheral T-cell lymphomas. http://1.usa.gov/1dvY1sd
Whereas efficacy of this combination was low in heavily pre-treated DLBCL, there was a signal of activity in relapsed/refractory PTCL utilizing the modified schedule.
* Am J Hematol 2013:
The aggressive peripheral T-cell lymphomas: 2013. http://1.usa.gov/1ic7OJB
References:
-
Treatment policy prepared by Dr. Kirsty Tompkins www.tsrcc.on.ca T-Cell.pdf
-
A Systematic Review and Meta-Analysis of Front-line Anthracycline-Based Chemotherapy Regimens for Peripheral T-Cell Lymphoma http://bit.ly/qFmamM
Return to top
Background Resources
PubMed queries on PCTL
.gif) |
Diagnosis | Review | Therapies | Prognosis
|
Clinical Trials
.gif) |
ClinicalTrials.gov studies for
Previously Untreated PTCL: http://bit.ly/f0LALB
Relapsed PTCL: http://bit.ly/dRSOak
|
.gif) |
The ASCO Post
Brentuximab Vedotin Shows Antitumor Activity in Patients With Relapsed Peripheral T-Cell Lymphoma http://bit.ly/1G6asLx
Among the 14 patients achieving an objective response, 8 had complete responses, including 5 patients with angioimmunoblastic T-cell lymphoma. At the time of the study report, the median duration of response for all patients was 7.6 months, with five responding patients remaining in follow-up and five on therapy.
This will lead to a study testing this agent as a part of first line therapy for this very challenging indication.
• PAL Query to help patients and physician locate such trials:
Studies for peripheral t-cell lymphoma testing Brentuximab Vedotin - List Results -
ClinicalTrials.gov http://1.usa.gov/1DGscZU
|
Treatment Updates
.gif) |
Summary of NCCN Guidelines 2011
See for details: nccn.org pdf (requires free subscription and login)
First-primary treatment
.gif) |
Clinical Trial preferred - Lookup:
Previously Untreated PTCL: http://bit.ly/f0LALB
|
.gif) |
CHOP for ALCL, ALK+ subtype
|
.gif) |
Other regimens to consider:
.gif) |
CHOP, then ICE
|
.gif) |
CHOP, then IVE
|
.gif) |
HyperCVAD
|
|
.gif) |
First-line consolidation (such as transplant)
.gif) |
All patients except low risk, or ALCL, ALK+ with good prognosis.
|
|
Second-line treatment (relapse setting) transplant eligible
.gif) |
Clinical Trial preferred - Lookup:
Relapsed PTCL: http://bit.ly/dRSOak
|
.gif) |
Transplant following:
.gif) |
DHAP / ESHAP / GDP / GemoOx / ICE / MINE
|
.gif) |
Pralatrexate (but not for AITL subtype)
|
.gif) |
Romidepsin
|
|
Second-line treatment (relapse setting) not eligible for transplant:
.gif) |
Clinical Trial preferred - Lookup:
Relapsed PTCL: http://bit.ly/dRSOak
|
.gif) |
Alemtuzumab (activity in small trial)
|
.gif) |
Bortezomib (activity in small trial)
|
.gif) |
Cylosporine for AITL subtype only
|
.gif) |
Denileukin Diftitox / Gemcitabine / Radiation therapy
|
.gif) |
Pralatrexate (but not for AITL subtype)
|
.gif) |
Romidepsin
|
|
.gif) |
Treatment policy prepared by Dr. Kirsty Tompkins www.tsrcc.on.ca T-Cell.pdf
|
.gif) |
Peripheral T cell lymphoma, not otherwise specified: the stuff of genes, dreams and therapies pubmedcentral.nih.gov/
(full text - technical)
|
.gif) |
Drug That Mimics Folic Acid
http://www.sciencedaily.com/releases/2008/12/081209155010.htm
n = 109 patients with peripheral t-cell lymphoma
"These results indicate that pralatrexate produces a major durable response in patients for whom numerous prior treatments have been unsuccessful," says Dr. Owen A. O'Connor
|
.gif) |
Autologous transplantation for relapsed or primary refractory peripheral T-cell lymphoma. http://bit.ly/yYvN9
"With a median follow-up time of 6 years for surviving patients with PTCL and DLBCL, the 5-year progression-free survival (PFS) rates for PTCL and DLBCL patients were 24% and 34% respectively (P = 0.14); the corresponding overall survival (OS) rates were 33% and 39% respectively. There were no significant differences between the two groups with respect to time to disease progression or survival after progression."
.gif) |
Also see Long-term results of autologous hematopoietic cell transplantation for peripheral T cell lymphoma: the Stanford experience. www.ncbi.nlm.nih.gov/pubmed/18541192
"Disease status at AHCT had a significant impact on PFS and OS. The 5-year PFS for patients in CR1/PR1, CR2/PR2+, and REF was 51%, 12%, and 0%, respectively, and the corresponding figures for OS were 76%, 40%, and 30%, respectively. The pretransplant factors that impacted survival were disease status and the number of prior regimens. Histology, age, sex, stage, B symptoms, bone marrow involvement, and duration of first response did not significantly affect PFS or OS."
|
|
.gif) |
Phase II trial of denileukin diftitox (Ontak) for relapsed/refractory T-cell non-Hodgkin lymphoma interscience.wiley.com
"Objective responses (six CRs, seven PRs) were achieved in 13 patients (48·1%), stable disease in eight (29·6%) and six (22·2%) had progressive disease. An objective response was achieved in eight of 13 patients (61·5%) with CD25+ tumours (four CR/four PR) and five of 11 patients (45·5%) with CD25− tumours (two CR/three PR). Median progression-free survival was 6 months (range, 1–38+ months). Most adverse reactions were grade 1/2 and transient. No grade 4–5 toxicities were reported. Denileukin diftitox had significant activity and was well tolerated in relapsed/refractory T-NHL, with responses observed in both CD25+ and CD25− tumours. Further studies of denileukin diftitox in combination with other agents are warranted in previously untreated and relapsed/refractory T-NHL."
|
.gif) |
Therapy with gemcitabine in pretreated peripheral T-cell lymphoma patients annonc.oxfordjournals.org
"Of the 13 patients, one achieved complete response (CR) and eight achieved partial responses (PR); the remaining four showed no benefit from the treatment. Among the responders, one CR and four PR were documented in the PTCLU patients and four PR in MF patients. Treatment was well tolerated; hematologic toxicity was mild and no nausea/vomiting or organ toxicity was recorded."
|
.gif) |
Review: Hematopoietic SCT for peripheral T-cell lymphoma?
Bone Marrow Transplant. 2008 Oct 20. PMID: 18936735
|
.gif) |
Gene expression analysis of peripheral T cell lymphoma, unspecified, reveals distinct profiles and new potential therapeutic targets jci.org (full text)
Notably, both phosphorylation of PDGFRα and sensitivity of cultured PTCL cells to imatinib (as well as to an inhibitor of histone deacetylase) were found. These results, which might be extended to other more rare PTCL categories, provide insight into tumor pathogenesis and clinical management of PTCL/U.
ClinicalTrials.gov
|
.gif) |
PTCL Prognostic marker: Expression of CYP3A4 as a predictor of response to chemotherapy in peripheral T-cell lymphomas. Blood. 2007 Nov 1;110(9):3345-51. Epub 2007 Jul 18. PMID: 17634410
|
.gif) |
Interim Response and Safety Analyses Support Continuation of PDX (pralatrexate) in Patients with Peripheral T-Cell Lymphoma www.earthtimes.org
Results of the interim analysis of patient response data exceeded the pre-specified threshold for continuation of the trial, which required a minimum of four responses (complete or partial) out of the first 35 evaluable patients, as determined by independent oncology review.
|
.gif) |
Frontline Autologous Stem Cell Transplantation (Asct) In High-Risk Peripheral T-Cell Lymphoma (PTCL): A Prospective Study From The Gel-Tamo Study Group blackwell-synergy.com
Conclusions: Early salvage therapy for patients with high-risk aggressive nodal PTCL that do not achieve CR after 3 courses of chemotherapy and ASCT frontline consolidation for chemosensitive patients may improve treatment outcome.
|
.gif) |
Long-term follow-up of patients with peripheral T-cell lymphomas treated up-front with high-dose chemotherapy followed by autologous stem cell transplantation.
Leukemia. 2006 Sep;20(9):1533-8. Epub 2006 Jul 27. PMID: 16871285
... our findings indicate (1) up-front high-dose therapy and ASCT are feasible, but could induce a high rate of long-term CR only in patients with ALK-positive ALCL and (2) the achievement of CR before autografting is a strong predictor of better survival."
|
.gif) |
Autologous transplantation for relapsed or primary refractory peripheral T-cell lymphoma.
Br J Haematol. 2006 Jun 6; PMID: 16759221
|
.gif) |
Marker Expression in Peripheral T-Cell Lymphoma: A Proposed Clinical-Pathologic Prognostic Score.
J Clin Oncol. 2006 Apr 24; PMID: 16636342
|
.gif) |
Gene expression profiling identifies molecular subgroups among nodal peripheral T-cell lymphomas.
Oncogene. 2006 Mar 9;25(10):1560-70. PMID: 16288225
|
.gif) |
Direct comparisons of peripheral T-cell lymphoma with diffuse B-cell lymphoma of comparable histological grades--should peripheral T-cell lymphoma be considered separately? jco.org
|
.gif) |
Stem cell transplantation for peripheral T-cell lymphomas.
Leuk Lymphoma. 2004 Mar;45(3):441-6. Review. PMID: 15160904
|
.gif) |
Peripheral T-cell lymphomas unspecified presenting in the skin: analysis of prognostic factors in a group of 82 patients. Blood. 2003 Sep 15;102(6):2213-9. Epub 2003 May 15. PMID: 12750155 | More
|
.gif) |
Peripheral T-cell lymphoma (excluding anaplastic large-cell lymphoma): results from the Non-Hodgkin’s Lymphoma Classification Project Annals of Oncology 13:140-149, 2002
|
.gif) |
Clinical features of peripheral T-cell lymphomas in 78 patients diagnosed according to the Revised European-American lymphoma (REAL) classification.
Eur J Cancer. 2002 Jan;38(1):75-81. PMID: 11750843 PubMed
|
.gif) |
Peripheral T-cell lymphoma [NK-type] Curr Oncol Rep. 2002 Sep;4(5):434-42. Review. PMID: 12162919 PubMed
|
.gif) |
Inhibitor of histone deacetylation, depsipeptide (FR901228), in the treatment of peripheral and cutaneous T-cell lymphoma: a case report. Blood. 2001 Nov 1;98(9):2865-8. PMID: 11675364 PubMed
|
In the News:
.gif) |
June 2015:
Belinostat for Relapsed or Refractory Peripheral T-Cell Lymphoma: Results of the Pivotal Phase II BELIEF Study http://bit.ly/1Ikqsf6
|
.gif) |
ASCO Post
Brentuximab Vedotin Shows Antitumor Activity in Patients With Relapsed Peripheral T-Cell Lymphoma http://www.ascopost.com
|
.gif) |
Blood 2014: Alison J. Moskowitz, Matthew A. Lunning and Steven M. Horwitz
How I treat the peripheral T-cell lymphomas http://bit.ly/1k8AW1Q
Technical -- but for anyone diagnosed with PTCL ... a very important paper to look over and share with our oncologists - underlining the potential importance of consulting specialists in this area.
One of the more challenging aspects in managing PTCL is confidently arriving at the diagnosis. T-cell lymphomas are rare, and we try to approach new diagnoses with at least a little skepticism. For many B-cell lymphomas, reproducible immunophenotypic patterns and cytogenetic features allow for an algorithmic approach to diagnosis. On the contrary, T-cell lymphomas are often characterized by antigen aberrancy that may vary within a subtype or even over the course of disease.7,8
The most powerful predictor of outcome in our series was interim PET; 53% of patients normalized their PET after 4 cycles of chemotherapy and in those who achieved interim PET-negative status, 59% were progression free at 5 years, including 53% of those with IPI of ≥3.
Our preference for fit patients is for alloSCT in the relapsed/refractory setting. Our institutional data and others have shown that the use of autologous stem cell transplantation for relapsed PTCL, with a possible exception of ALCL, has rarely resulted in long-term disease control.40,41 This is somewhat controversial and a recent analysis from the Center for International Blood and Marrow Transplant Research registry points to better results with ASCT at relapse, although the series is overrepresented by ALCL including many with ALK-positive ALCL.42
|
.gif) |
Novel therapies for peripheral T-cell lymphomas (full text) ncbi.nlm.nih.gov
Accumulating data indicate that these novel agents have little cumulative toxicity and can be administered continuously to patients who are not candidates for consolidative stem-cell transplantation (SCT), with little impact on quality of life. They might also provide a new salvage option for patients eligible for SCT with no impact on autologous stem-cell collection or subsequent engraftment.
|
.gif) |
Oncology Times: PTCL: Should Initial Management Include High-Dose Therapy with Autologous Stem Cell Rescue?
|
Return to top
|