|
Putting the ClinicalTrials.gov registry to
work for patients, oncologists, and scientists
 PAL embraces science- and evidence-based medicine as the only reliable path forward to improve outcomes for patients - present and future. PAL embraces science- and evidence-based medicine as the only reliable path forward to improve outcomes for patients - present and future.
The patient has one life to experiment with; and dependents to consider. Thus the decision to participate in a trial will be based on the merits of doing so as a treatment decision - not to advance the science or to help win marketing approval for a study drug.
Therefore, the ideal clinical trial is a marriage of the best science, rigorous study methods, and high-quality medicine.
For a clinical trial to enroll patients efficiently it must:
1) Compare well to regular treatments -
or competing studies - as it relates to the
patient's clinical circumstance and treatment goals;
See also 7 Reasons to Consider Clinical Trials based on our Clinical Circumstances
2) The referring physician or patient must be aware of it --
it's location, eligibility criteria, and have good confidence in the supporting science and evidence to date.
To address item 2, PAL provides queries of ClinicalTrials.gov
... in various ways in multiple venues:
- Through our website
- By posts made to our support forums
- By distribution notices made to social media – Facebook and Twitter.
We have developed three main approaches to finding trials listed here and illustrated below:
1. Following an encouraging published report,
by querying ClinicalTrials.gov for open trials by the study agent
2. With Trial-finding queries for the type of lymphoma and the patient’s
treatment status (untreated, relapsed, other)
3. By listing PAL’s Picks – Trials of Interest
(grouped by type of lymphoma, in study drug order)
NOTE: PAL's Picks are studies that look promising to our group, vetted by our scientific advisors, based on recent reports. We list these in order to foster the conversation about trials between patients and their treating physicians - not to promote or match a patient to a particular trial.
PAL is committed to helping the community to appreciate how study design influences the confidence that we can have in the findings. We do our best to avoid hyping or promoting specific approaches to treatment.
4. By providing a list of independent experts to consult as part of a second opinion:
Trial Talk Experts
Find Trials By AGENT
such as for kinase inhibitors
Kinase inhibitors
Inhibit pathways that are overactive in cancer cells
|
See also Agents that target disease pathways |
B-cell Receptor pathway
|
|
Target
|
|
|
|
Reports | Counts |
AVL-292
|
B |
BTK |
- |
Find trials |
98 |
Reports
>
|
AVL-292
|
B |
BTK |
- |
Find trials |
98 |
Reports
>
|
Idelalisib / GS-1101 / CAL101
|
B |
PI3k delta |
- |
Find trials |
529 |
Reports
>
|
From this page the community can:
.gif) |
· Click name of agent to list articles on Google scholar.
|
.gif) |
· Click Find trials to list files for this agent in the ClinicalTrials.gov registry.
|
.gif) |
· Click the > symbol to see latest click count by bitley.
|
.gif) |
· Click Reports to list published outcome reports on the agent on Google Scholar
|
Find Trials By TYPE of LYMPHOMA and TREATMENTS status
Here the QUERIES can help you to list only clinical trials for
your type of lymphoma AND treatment status - such as previously untreated follicular lymphoma.
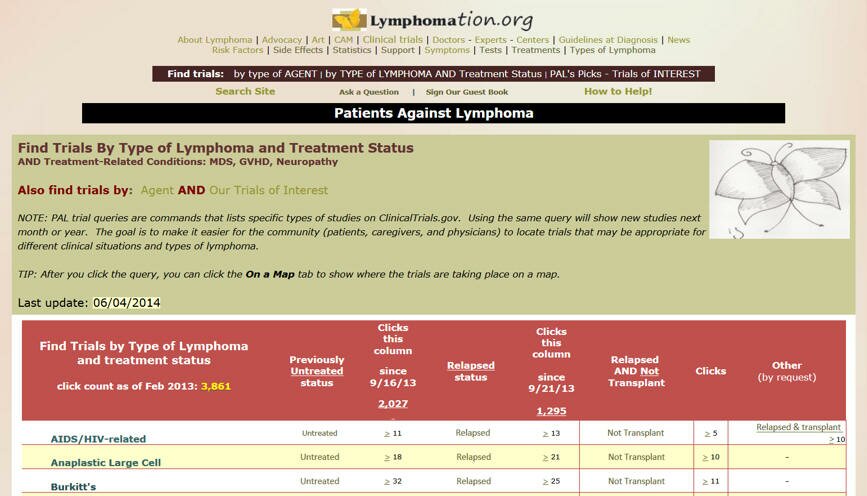
From this page the visitor can:
.gif) |
· Click the type of lymphoma for background on it, such as its natural history.
|
.gif) |
· Click Untreated or Relapsed to list studies in the ClinicalTrials.gov registry based on the diagnosis and treatment status.
|
.gif) |
· Click the > symbol to see latest click count by bitley.
|
These are studies that look promising to our group and by our scientific advisors based on recent reports. We list these in order to foster the conversation about trials with the patient’s oncologist or by consulting an expert for a second opinion. We are very careful not to recommend participation in any specific trial.
Open PDF explaining how to use this page
OPTIONS FROM CLINICALTRIALS.GOV
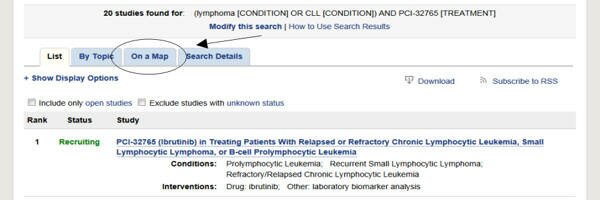
After clicking some queries, you will see a list of studies.
.gif) |
You can then click the ON A MAP tab
to find the locations of the studies on a map.
|
.gif) |
You can also click a check box to show only open studies
|
.gif) |
Click the protocol name to show the details of the study, such as Contacts and Locations and Eligibility
|
Supporting efficient research and clinical decision-making
.gif) |
Patients and physicians must know what trials are running in order to consider them. How else?
|
.gif) |
Our tools may also be useful to investigators in order to judge interest in agents and to know what is being studied to guide in the design of their trials.
|
Our Tools are Possible by Community Support
Our tools, and the related educational projects would not be possible without the generous support of the patient community. So we are taking time here to thanks all who have donated and intend to do so soon.
PAL's continuing existence truly depends on your help.
While the industry investment is essential in order to bring new drugs to market, we have maintained our financial independence so that you can be assured that a financial interest is not guiding the content we provide.
Clinical Trial Tools At-a-Glance
|